Ingredients
INGREDIENTS OF SPRAY AWAY D.O.A.
The formulation of Spray Away D.O.A. is patent pending.
THREE INGREDIENTS: There are three safe ingredients in Spray Away when used according to instructions. Alcohol is common to most sanitizers. We used 62% denatured ethanol. It is not commonly communicated that alcohol’s anti-microbial effect lasts only when wet. After evaporation, the surface sprayed should be free of microbes, but there is no lasting protection.
What is unique about Spray Away D.O.A. is the protocatechuate crystals in the solution, upon the evaporation of the alcohol leaves a uniform crystalline anti-microbial coating. This gives a long-lasting anti-microbial effect on the hands, masks, or hard surfaces. Your skin is protected by the healing properties of the coating.

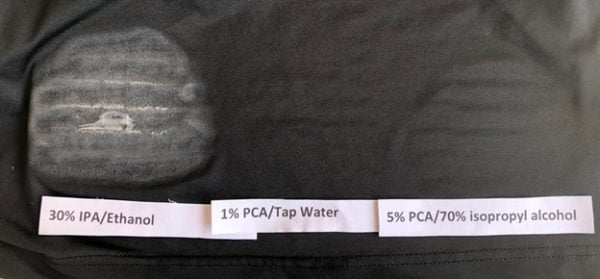

Protocatechuate is food substance. It is common to the human diet, found in many foods. It is the primary metabolite upon eating of blueberries or cherries. It is classified as a nutrceutical as opposed to a pharmaceutical; i.e. drug. It is scientifically a phytochemical, biochemically produced in our application rather than per plant extraction. Protocatechuate even though a nutraceutical by classification has been researched and developed to meet FDA device and or drug regulations. It is as grey/white crystal of 177 microns or 80 mesh size.

The protocatechuate crystals in solution have sharp needle like shapes which produce the initial anti-microbial function by physically disrupting the microbe.
The protocatechuate crystals are easily identified in solution under polarized light microscopy.
A fragrance is common to most sanitizers. We used limonene.
There are three ingredients in the Spray Away D.O.A. sanitizer:
Denatured Ethanol 62%:
This concentration is FDA approved for use in a sanitizer. Denatured ethanol is not the ethanol found in alcoholic beverages. Do not drink and keep away from mouth or eyes.
Protocatechuic Acid 1.0%:
This is a nutraceutical. The FDA has listed this as Generally Recognized As Safe (G.R.A. S.) as a food flavoring. This is safe to eat when used with foods, but not in this application. There is evidence it has an anti-microbial property as seen in the following US patents. [see list below] After the list put in Wash Away info: Wash Away D.O.A. is mostly sterile water. There is 1% PCA no other additive. It is an adjunct to mitigation of areas of the nose and oral pharynx.
Lemonene 0.6%:
This is the oil found in citrus fruits. In manufacturing, limonene is used as a fragrance, cleaner (solvent), and as an ingredient in water -free hand cleansers. It is a fragrance in Spray Away D.O.A.
The following US Patents have been issued to Dr. Johnson concerning the many applications of protocatechuic. They have resulted not only in Spray Away but also in many other health applications.
September 11, 2012. This patent showed that the dyes of plants (anthocyanins and anthocyanins) the precursor of the metabolite protocatechuic acid turned on the gene for growth hormone IGF-1 in human synovium.
November 8, 2016. This patent secured the intra articular injection route not previously included in the granted patent claims for cyanidin-3-glucoside.
November 22, 2016. This was the parent patent showing broad spectrum antibiotic properties and wound healing acceleration. See the illustrations above.
March 27, 2018.This was a continuation of the parent application that adds 2,4,6 trihydroxybenzaldehyde to the broad-spectrum antibiotic, plus collagen proliferation (i.e. wrinkle treatment) and reduction in scar formation.
March 27, 2018.This was a continuation of the parent application that adds 2,4,6 trihydroxybenzaldehyde to the broad-spectrum antibiotic, plus collagen proliferation (i.e. wrinkle treatment) and reduction in scar formation.
May 15, 2018. This patent secures that protocatechuic acid can only be manufactured and delivered to an end user by the owner of this patent.
June 5, 2018. This patent was a continuation of 9,925,152 that now allowed for intra-articular injection.
July 26, 2018. This patent included the metabolites of protocatechuic acid and 2,4,6 trihydroxybenzaldehyde for the destruction of biofilms of MRSA and Pseudomonas on metal, cloth and ceramic.
July 26, 2018. This was an expansion of 10,004,705 applications.
July 10, 2018. This patent expanded the formulation for treatment of implants to include protocatechuic acid, 70% isopropyl alcohol, propylene glycol, and an essential oil.
July 31, 2018. This patent includes the chemical formulation of 10,016,380 for medical and surgical implants, dental implants and instrumentation. The later for spaying or soaking in solution.
December 4, 2018. This patent secures the use of PCA to coat a bandage or dressing at time or treatment and or a commercial preparation for as stated in claim #22 if a wound is a burn, skin break, bone break, muscle tear, puncture, surgical incision site, microdermabrasion site, skin graft site, a wound associated with diabetes, a bed sore, a pressure sore, skin defoliation, or a laceration, and wherein the protocatechuic acid becomes activated by contact with moisture from the wound.
April 23, 2019. An extension of prior claims that specifically cite PCA as an antibacterial for Propionibacterium acnes. This for the treatment of skin acne.
May 21, 2019. This patent extends the use of PCA to the food industry.
September 3, 2019. This is a non-surgical method of loosening biofilms from an implant via ultrasound and then treating without surgery. The treatment would be repeated needle aspirations for infection status and then injection of protocatechuic acid crystals to destroy the biofilms attached to the implant.
October 1, 2019. This is an extension and further specification of the skin penetration formulation claims for pre-operative skin disinfection and facial acne with concentration above 10%.
September 5, 2020. This is an extension that includes the disinfectant and sanitizer use emphasizing the residual anti-microbial protocatechuic acid coating the remains on the article and or person after the evaporation of the liquid vehicle.
March 30, 2021. The treatment of COVID19 virus with protocatechuic acid. This patent is for treating the patient; oral, intravenous, injection, etc.
I claim:
1. A method of treating Covid-19 in a mammal comprising: administering to the mammal in need thereof a composition comprising protocatechuic acid.
2. The method of claim 1, wherein the mammal is a human.
August 31, 2021. Anti-microbials and the Methods and Use thereof. This patent is for mitigation of bacteria and virus (COVID 19) by coating of personal protective equipment; masks, gowns, hats, shoes, etc.
3/8/2022. Anti-microbials and the Methods and Use thereof. This patent for the mitigation of SARS Co-2 virus on skin, protective equipment and hard surfaces.
I claim:
1. A method of interrupting the transmission of SARS CoV2 virus comprising: spraying, coating, fogging, and/or infusing a subject’s skin or personal protective equipment, room and/or facility, with a composition comprising protocatechuic acid, a liquid vehicle comprising an alcohol, and a stabilizer, the composition disinfecting SARS CoV2 virus on the subject’s skin or personal protective equipment, room and/or facility, upon contact; and forming a solid coating of protocathechuic acid on the subject’s skin or personal protective equipment, room and/or facility upon evaporation of the liquid vehicle, and disinfecting SARS CoV2 virus with the solid coating of protocatechuic acid for up to 24 hours.
May 24, 2022. Candida auris disinfectant.
May 24, 2022. Compositions including combination of cannabinoid (CBS) and protocatechuic acid.



